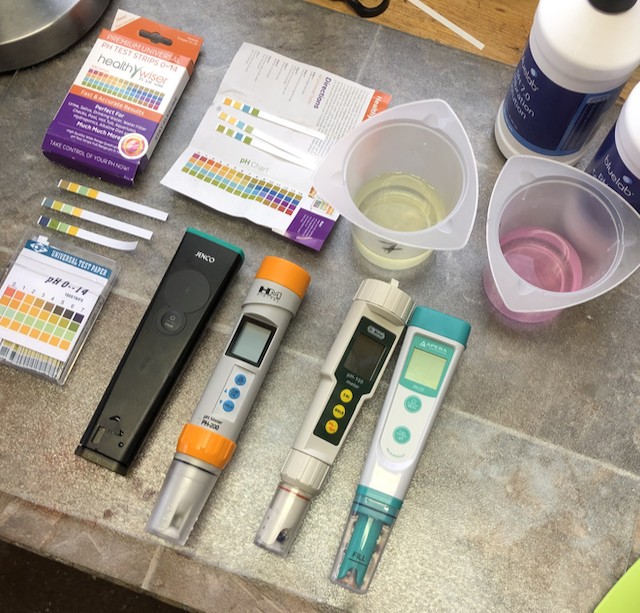
Tips & techniques: You don’t need to test and alter the pH of an emulsion with a few exceptions…
Welcome back to another instalment of tips & techniques! I thought I should create a separate and updated post on this topic because I see people going through all the work to test and alter pH for emulsions that are just fine on their own. As you’ll see, most emulsifiers are designed to create formulas...